Because Oqton is such an open platform, it allows you to configure it to meet the user's needs
Bert Engelen
Quality Manager at Amnovis
Based in Aarschot, Belgium, the company was founded in June 2020. “We're a start-up engineering and production company that uses additive manufacturing to make high-end products for quality critical applications such as medical devices”, explains Ruben Wauthle, CEO and one of the company's founders. “As a company, we want to enable innovations in additive manufacturing by introducing new additive technologies, materials, and applications at a production level and help our customers bring new products to the market.”
Amnovis offers best-in-class metal powder bed fusion and post-processing, with an eye for emerging additive manufacturing technologies and materials. “We saw a lot of challenges in the market”, says Wauthle, “but at the same time, we also saw opportunities to do things better and faster”. This included identifying huge potential for introducing new additive technologies, materials, and applications and producing conforming parts to customer specifications. Amnovis quickly established its USP as “driving innovation for AM contract manufacturing of high-end products, with a huge focus on quality and being able to deliver on time.”
Key to meeting these objectives was a cloud-based system. “We started the company with a strong focus on flexible and scalable operations”, explains Wauthle, who started his career at LayerWise (now 3D Systems) in 2010 as Medical Application Engineer, obtaining his PhD in Mechanical Engineering in 2014 from KU Leuven. “We decided to go completely digital and paperless. Our entire production and quality management system is completely paperless. We're very proud of that.”
This led Amnovis to partner with Oqton from the outset. “We decided to partner with Oqton while we were in the process of founding the company”, he says. “We had a demo of the software and were amazed by the capabilities and the potential of the software platform.” In addition, he found the Oqton team “very responsive” and willing to listen and react to feedback.
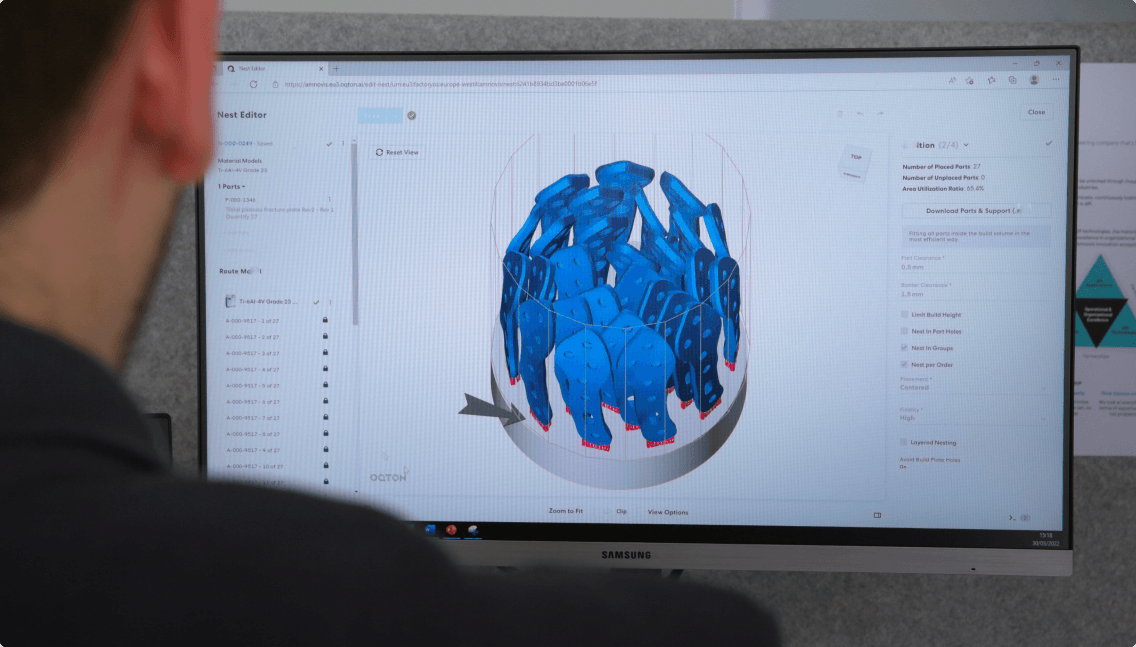
Once operational, “what amazed us the most about the Oqton software is the huge amount of data that it captures”, enthuses Wauthle, especially in terms of traceability from raw material to finished product. “We can capture everything in a digital form”, he says. “It's all centralized. And on top of that, the software architecture is set up in such a way that you can really use the data, you can pull up reports, analyze the data, and look for trends in your production process.” Designed specifically for additive manufacturers, Oqton “is the perfect fit”.
To achieve the industry best standard, the next step for Amnovis was to obtain ISO 13485 certification. Amnovis did so in August 2021, a little more than a year after its founding. Bert Engelen, Quality Manager at Amnovis, explains that ISO 13485 certification “is all about the quality management system that you set up and how that ensures the quality, safety, and performance of the medical devices that you manufacture.” Obtaining ISO 13485 “really put us on the radar of many of our potential customers”, explains Engelen. “It has already sparked some interesting discussions and some interesting projects that we are currently working on.”

Engelen – who started as Project Engineer in the Healthcare department at LayerWise (now 3D Systems) – attributes achieving ISO 13485 so quickly to working with Oqton software from the start. “Oqton really is the backbone of our operations”, he explains. “It's the single source of truth for all information related to the products that we make: every material, every piece of equipment, every tool that we have and operate, Oqton holds the information.” This includes calibration, maintenance schedules, maintenance records, full traceability, and revision control as products or customer requirements change.
In particular, this makes for a smooth audit process. Engelen describes audits as an integral part of the industry. Customers that want to manufacture their products with a new provider typically audit at least once before they start a product manufacturing partnership. Follow-up audits, as well as unannounced audits, are then part of the day-to-day routine.

Because Oqton is such an open platform, it allows you to configure it to meet the user's needs
Bert Engelen
Quality Manager at Amnovis

Amnovis customers include CairnSurgical for the additive manufacturing of its revolutionary Breast Cancer Locator ™ (BCL) for patients in Europe. The BCL is a patient-specific surgical guidance solution designed to assist surgeons in locating and accurately excising breast tumors. Amnovis' founders were also among the first to deploy L-PFB for printing titanium medical devices, such as orthopedic and spinal implants.
Amnovis has the additive manufacturing expertise to meet the need for higher speed and flexibility, while Oqton empowers this aspiration with live dashboards and automated reports for every step in the additive manufacturing process, including post-processing. Oqton gives a complete overview of your facility and boosts productivity.

The team at Oqton has been very instrumental in guiding us. They understand our needs and match the platform functionality to meet those needs.
Ruben Wauthle
Amnovis CEO
When the production workflow is up and running, he adds, “it is very intuitive for a new operator to understand, with little to no training: they can find all the information that they need in the platform and register the actions that need to be recorded.”
Amnovis are now widely viewed as an additive manufacturing trailblazer. With a market track record dating back to 2008, this independent and privately owned company is built on the strong foundation of long-term partnerships. Its ISO 13485 certification further underlines its ability to meet demanding, internationally agreed quality management standards.
“Oqton has very quickly become an invaluable tool for our day-to-day operations”, says Wauthle. But, he adds, this is only the start. “I am convinced that we're still only leveraging a small part of the true capabilities of the platform today. I know that future developments will allow us to integrate operations into the Oqton platform even further.” Together, Amnovis and Oqton will continue paving the way for the use of emerging additive manufacturing technologies and materials well into the future.

Founded in June 2020 by Peter Mercelis and Ruben Wauthle and based in Aarschot, Belgium, Amnovis is a manufacturing and engineering company that uses innovative additive manufacturing technologies and materials for high-end applications such as medical devices. As an ISO 13485 certified contract manufacturer, Amnovis offers best in class metal powder bed fusion and post-processing. Whichever technology and material fit an application – be that conforming parts or on time - that's what Amnovis' production is all about.